
Image result for michaelis menten graph Reaction Rate, Biochemical, Biochemistry, Line Chart
Km is the Michaelis-Menten constant, in the same units as X. It is the substrate concentration needed to achieve a half-maximum enzyme velocity. Create a Lineweaver-Burk plot. Before nonlinear regression was available, investigators had to transform curved data into straight lines, so they could analyze with linear regression. One way to do.

Illustration of MichaelisMenten function. Download Scientific Diagram
The NLFit dialog is an interactive tool which allows you to monitor the fitting procedure during the non-linear fitting process. This tutorial fits the Michaelis-Menten function, which is a basic model in Enzyme Kinetics, and shows you some basic features of the NLFit dialog. During the fitting, we will illustrate how to perform a Global Fit.

2 Model MichaelisMenton plots showing predicted changes with... Download Scientific
Page ID. Two 20 th century scientists, Leonor Michaelis and Maud Leonora Menten, proposed the model known as Michaelis-Menten Kinetics to account for enzymatic dynamics. The model serves to explain how an enzyme can cause kinetic rate enhancement of a reaction and explains how reaction rates depends on the concentration of enzyme and substrate.

The MichaelisMenten diagram of soluble and immobilized form of DHODH... Download Scientific
Michaelis and Menten equation (MM equation) has dominated biochemistry for more than a century after its seminal introduction in a paper published in 1913 in the journal Biochemische Zeitschrift, a predecessor of FEBS Journal [[]].Hence, publishing this guide in FEBS Journal would represent an apt dedication to the unmatched service rendered by this journal to the scientific community for more.

MichaelisMentenGleichung… BASICS of ANESTHESIOLOGY boa.coach
The Michaelis constant is the substrate concentration at which an enzyme operates at one half of its maximum velocity. How can you calculate this constant ba.

Developing a three‐dimensional animation for deeper molecular understanding of michaelismenten
The Michaelis-Menten equation (see below) is commonly used to study the kinetics of reaction catalysis by enzymes as well as the kinetics of transport by transporters. Typically, the rate of reaction (or reaction velocity) is experimentally measured at several substrate concentration values. The range of substrate concentrations is chosen such that very low reaction rates as well as saturating.
Physikalische Chemie
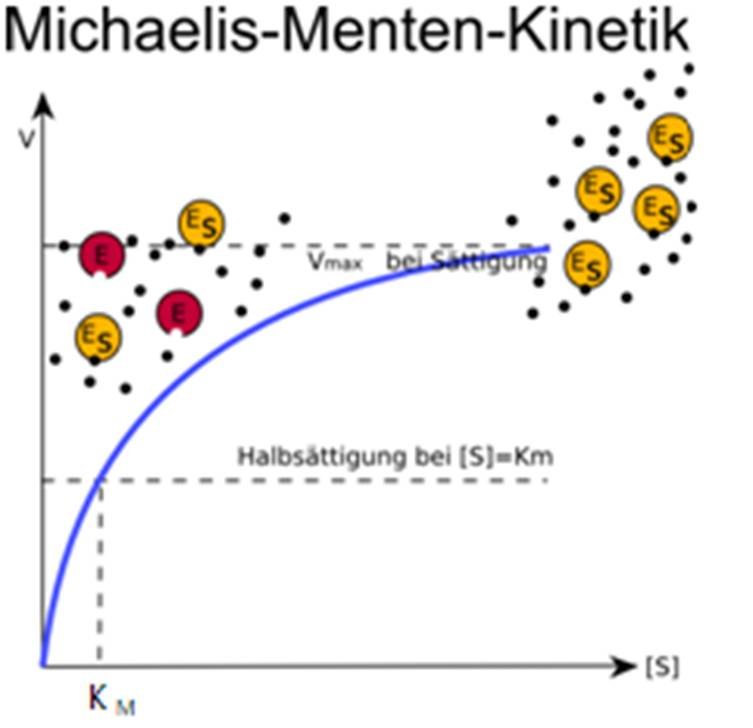
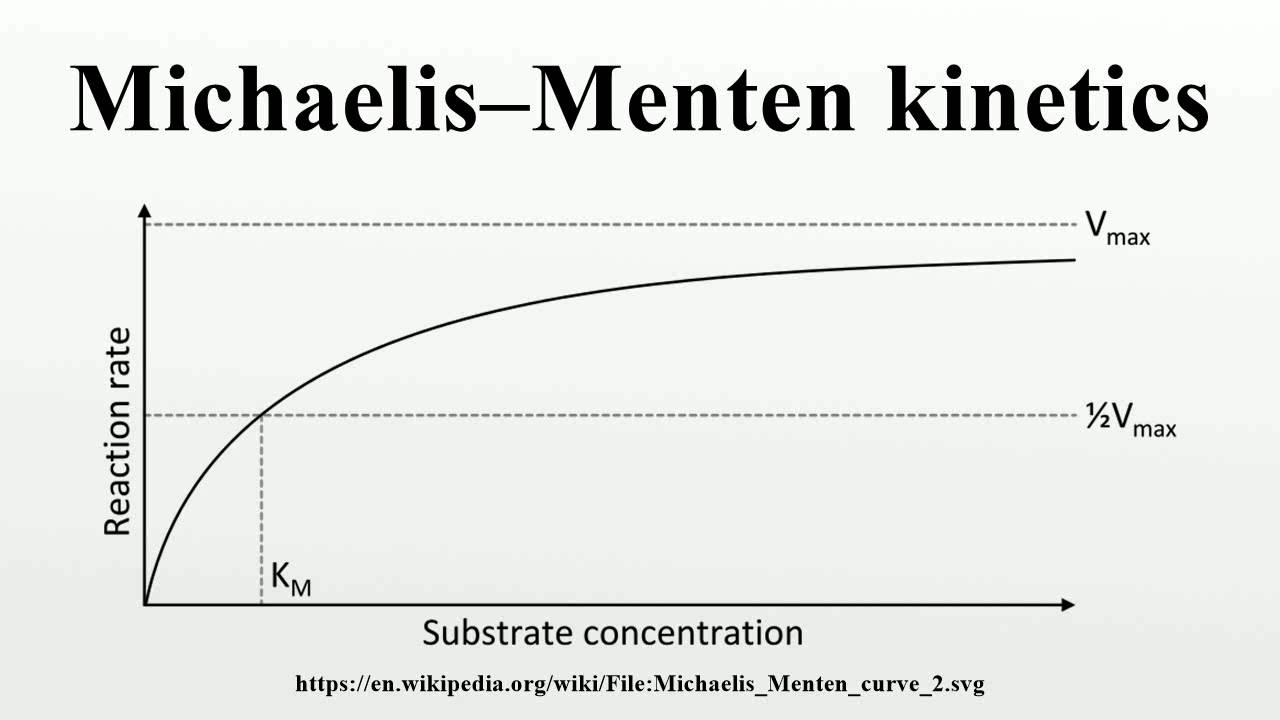
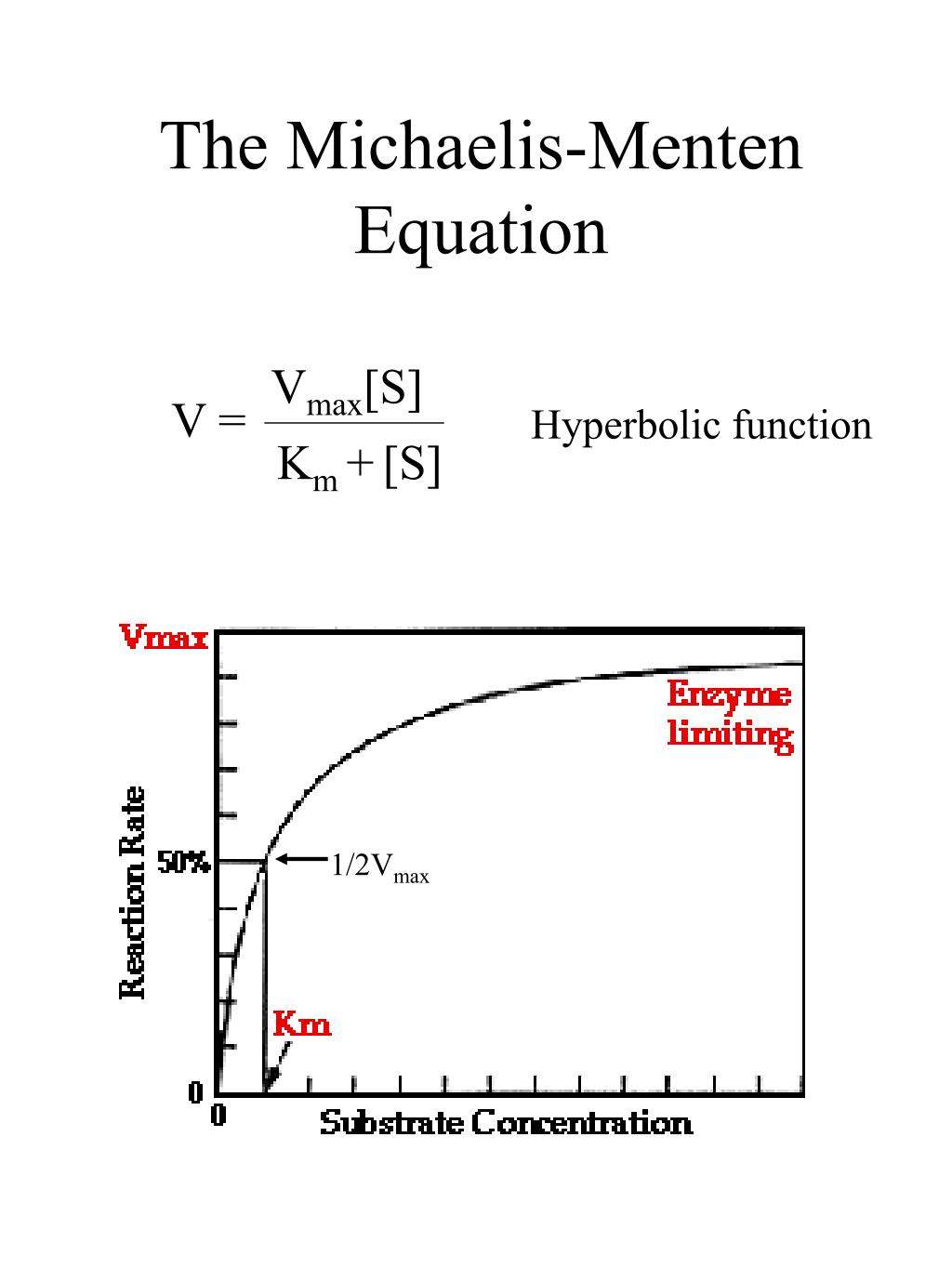
The reaction is zero-order kinetics. Figure 5.3.2: Diagram of reaction velocity and Michaelis-Menten kinetics. v = Vmax 2 = Vmax[S] Km + [S] (5.3.17) (5.3.17) v = V m a x 2 = V m a x [ S] K m + [ S] Therefore, Km K m is equal to the concentration of the substrate when the rate is half of the maximum velocity.

MichaelisMenten enzyme for TMT1 (green diamonds ), TMT2... Download Scientific Diagram
Michaelis-Menten kinetics. In biochemistry, Michaelis-Menten kinetics, named after Leonor Michaelis and Maud Menten, is the simplest case of enzyme kinetics, applied to enzyme-catalysed reactions of one substrate and one product. It takes the form of an equation describing the reaction rate (rate of formation of product P, with.

grafico MichaelisMenten Bald Mountain Science
This short video shows you how to set up the Michaelis-Menten model in Excel to model the creation of product as a function of substrate concentration.

MichaelisMenten study of the nonenzymatic RNA copying rates of 2AIpG... Download Scientific
STEP 3: Plot Michaelis Menten Graph with Calculated Velocity. To plot the graph, you need to select the values of Concentration and corresponding Velocity. Here, we have selected the range B4:C14. After that, go to the Insert tab and click on the Insert Scatter icon. A drop-down menu will appear.

The MichaelisMenten Enzyme Model
In this article we will discuss about the Michaelis-Menten Constant and Significance of Michaelis-Menten Constant.. Michaelis-Menten Constant: In an enzyme catalysed reaction when there is large excess of substrate and the enzyme concentration is held constant, if substrate concentration (S) is plotted against velocity (V) or reaction rate, a hyperbolic curve is obtained (fig. 10.13).
MichaelisMenten YouTube
The Michaelis-Menten equation, named after biochemist Leonor Michaelis and physician Maud Menten, "describes the relationship between the rate of substrate conversion by an enzyme (V) and the concentration of the substrate ([S])," according to the Davidson College Chemistry website. Based on this equation, the Michaelis-Menten curve can be.
PPT LAB 3 Enzyme PowerPoint Presentation, free download ID4526880
Named after Leonor Michaelis and Maud Menten, the Michaelis-Menten kinetics is the most basic form of enzyme kinetics. The equation is given by: \ ( v = \frac {d [p]} {dt} = \frac {Va} {K_m + a} \) Advertisements. In this equation, V or often represented as Vmax , signifies the maximum rate achieved by the system when the substrate.
MichaelisMenten plot for enzymesubstrate Download Scientific Diagram
The program determines the constants Rmax and Km of the Michaelis-Menten model using data given in Fundamentals of Chemical Reaction Engineering by M. E. Davis & R. J. Davis, McGraw Hill, 2003. This data represents the substrate (catechol) concentration versus time. This data was obtained using E. herbicola immobilized in a polymer gel to.

MichaelisMenten MichaelisMenten plot of MpIspS initial... Download Scientific Diagram
In biochemistry, the direct linear plot is a graphical method for enzyme kinetics data following the Michaelis-Menten equation. [1] In this plot, observations are not plotted as points, but as lines in parameter space with axes and , such that each observation of a rate at substrate concentration is represented by a straight line with.

Example of a MichaelisMenten plot (left) and a LineweaverBurk plot Download Scientific Diagram
The Michaelis-Menten mechanism (Michaelis & Menten, 1913) is one which many enzyme mitigated reactions follow. The basic mechanism involves an enzyme ( E, a biological catalyst) and a substrate ( S) which must connect to form an enzyme-substrate complex ( ES) in order for the substrate to be degraded (or augmented) to form a product ( P ).