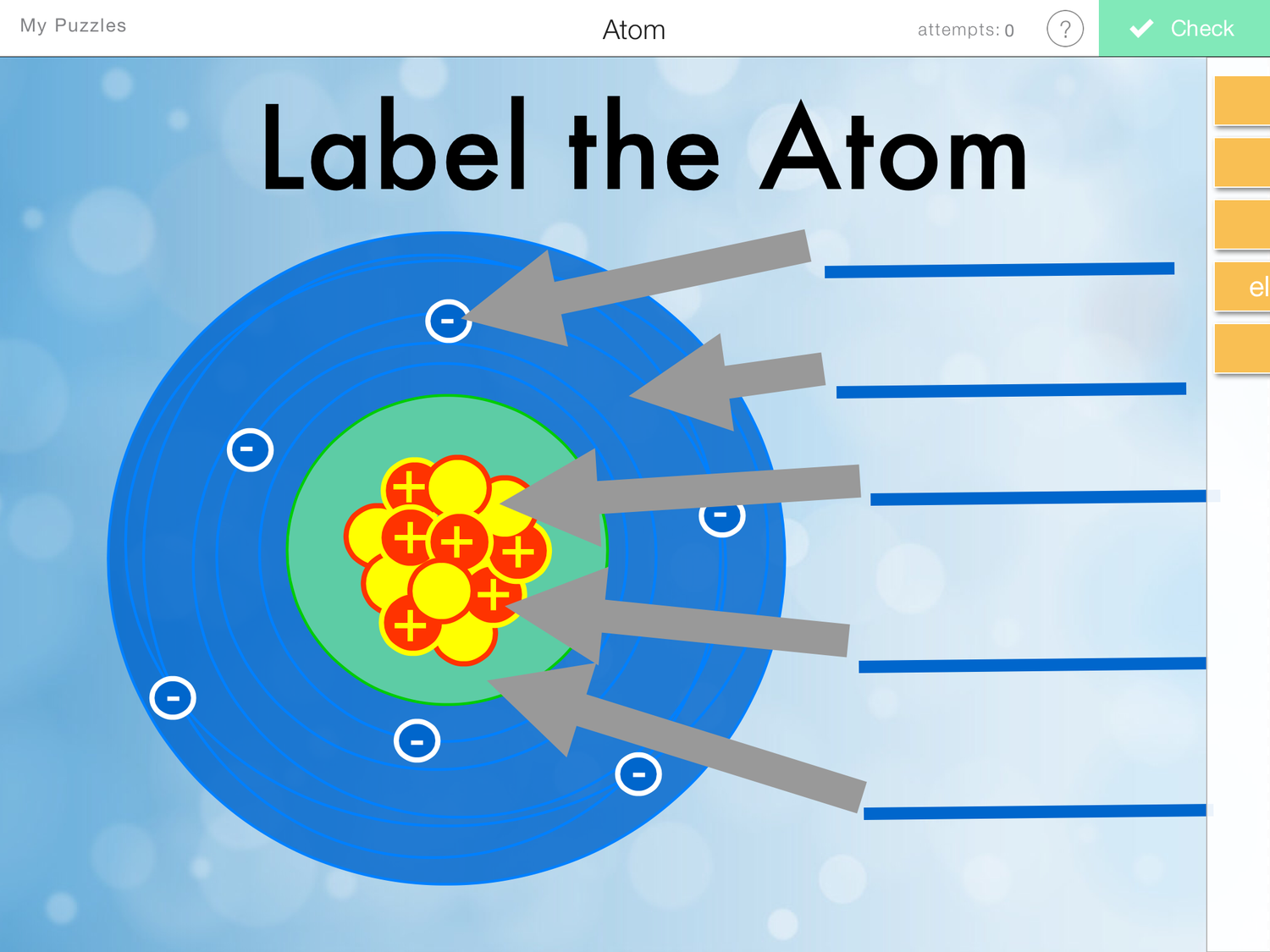
Label Parts of an Atom — Learning in Hand with Tony Vincent
All atoms are roughly the same size, whether they have 3 or 90 electrons. Approximately 50 million atoms of solid matter lined up in a row would measure 1 cm (0.4 inch). A convenient unit of length for measuring atomic sizes is the angstrom (Å), defined as 10 −10 metre. The radius of an atom measures 1-2 Å.
What is an Atom? Definitions & Examples Let us learn Basics News Bugz
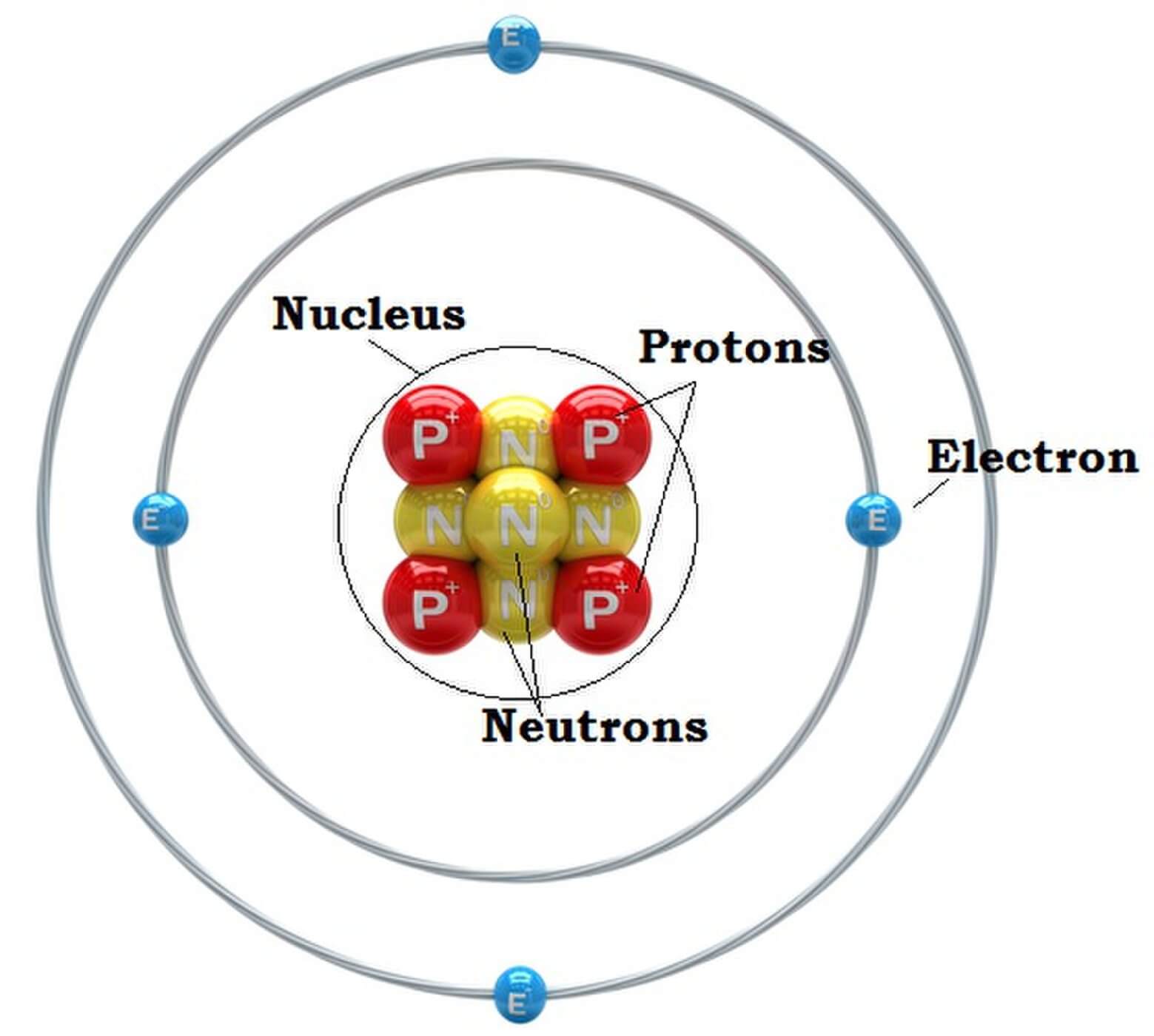
The Structure of an Atom: Parts, Diagram, Examples Anything that has mass and occupies space is called matter. The matter is made up of atoms. Atomic structure is the structure of an atom that consists of a nucleus (the centre), protons (positively charged), and neutrons (neutral).
Lets Get Inside An Atom!! The Science Station
Figure 2.2.1 2.2. 1: The Structure of the Atom. Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus. The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different.
Atoms, Molecules, and Compounds What's the Difference? Owlcation
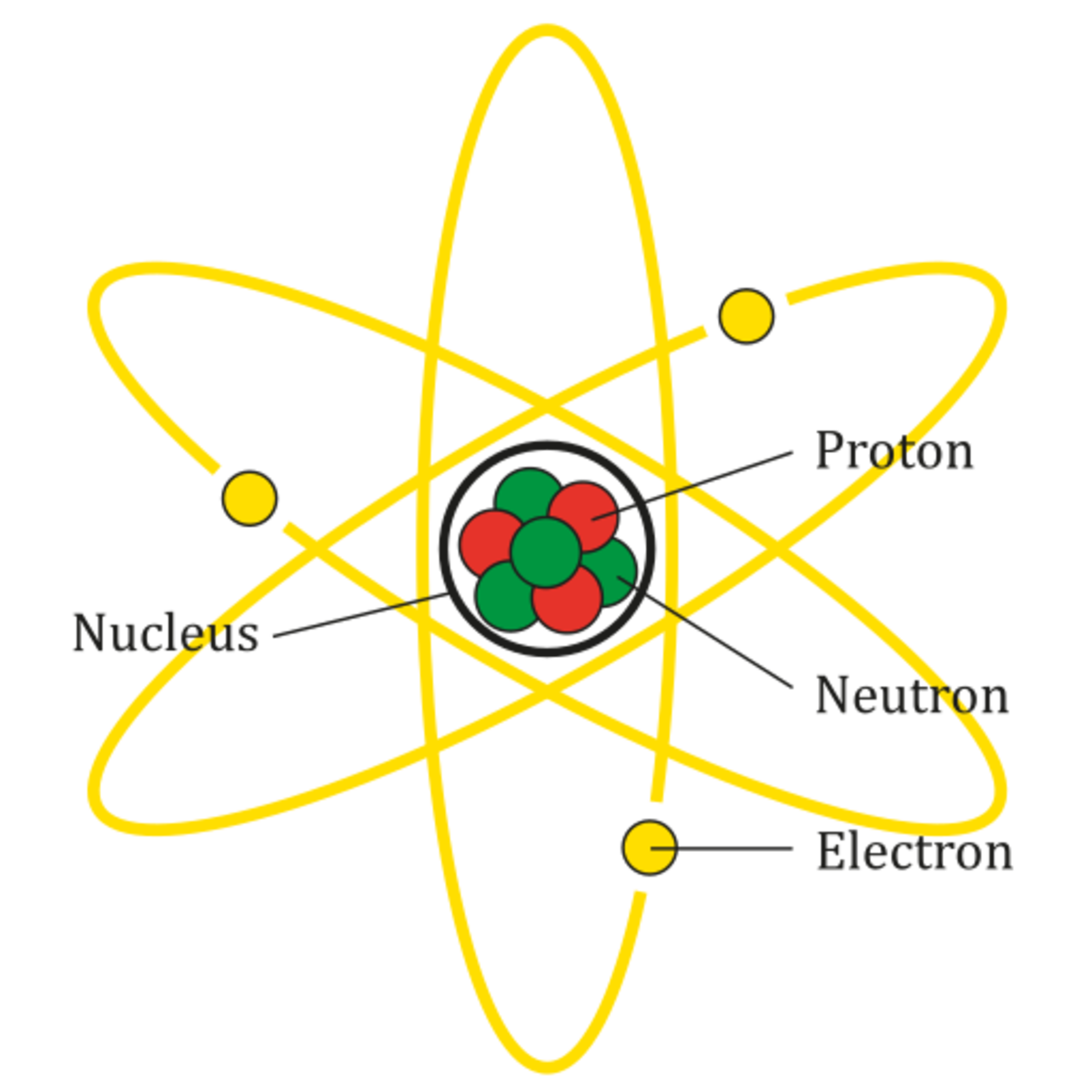
The Bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, similar to planets orbiting around the sun.
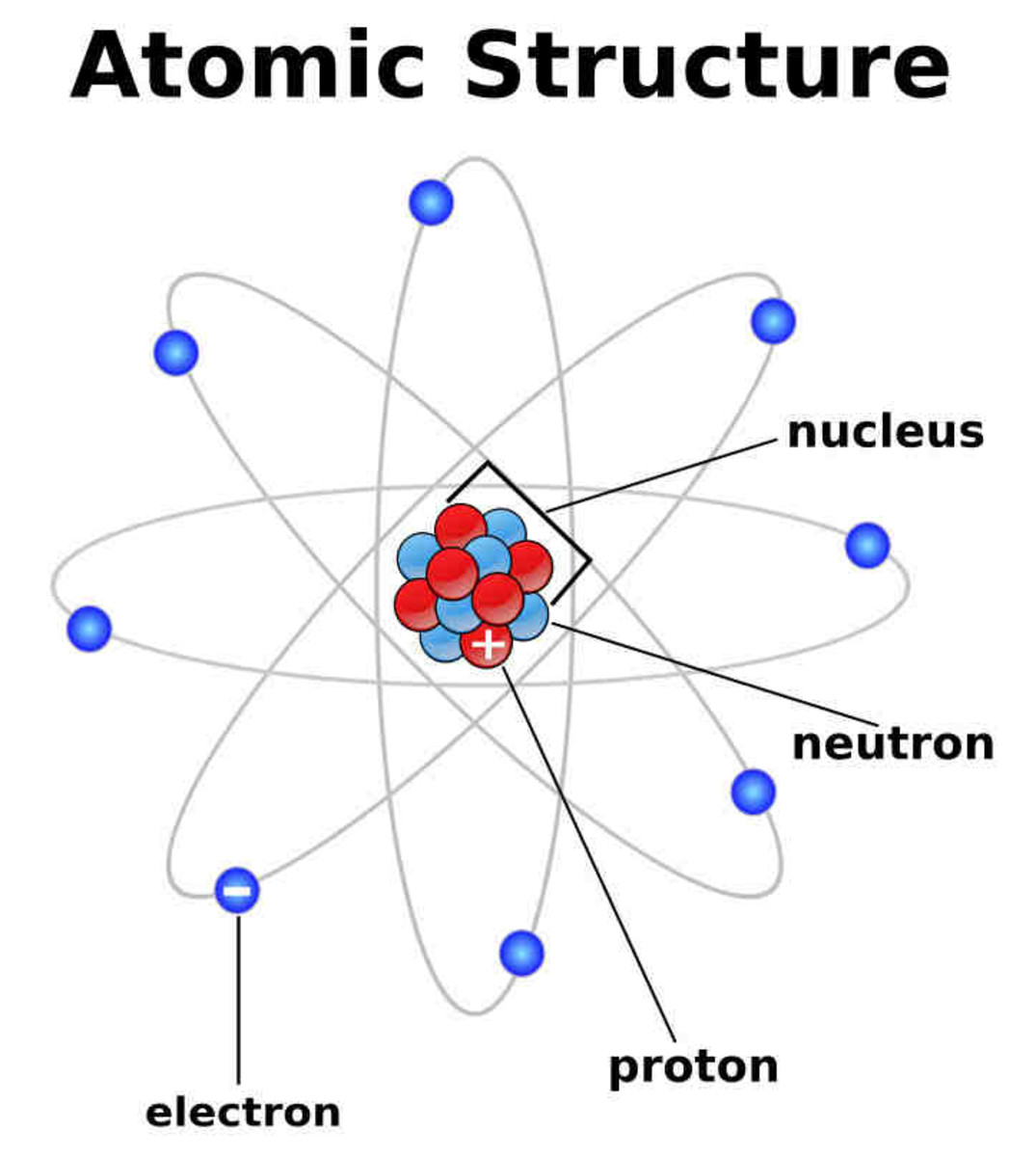
Atoms and Atomic Structure HubPages
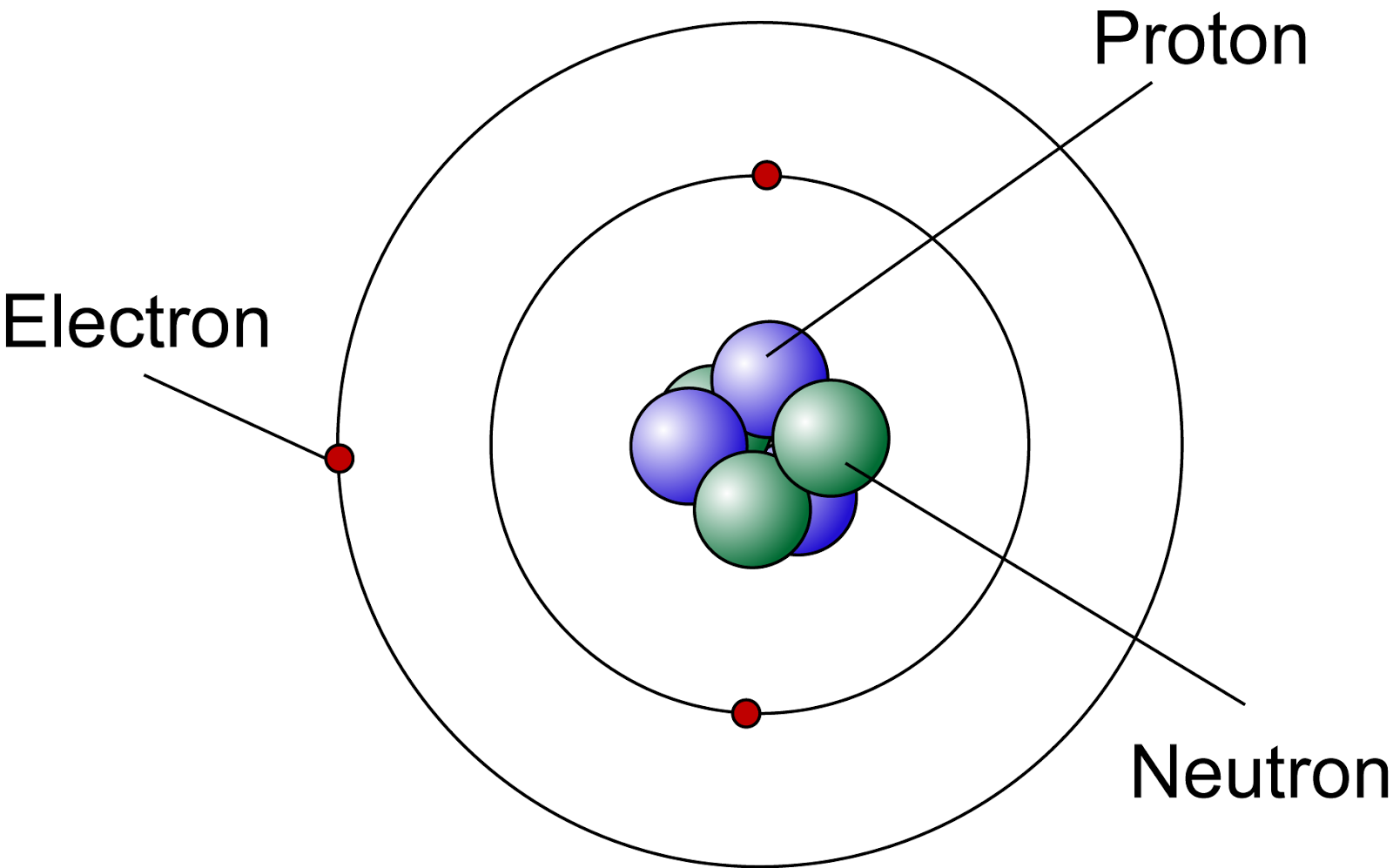
Atom The atom is the basic particle of the chemical elements. An atom consists of a nucleus of protons and generally neutrons, surrounded by an electromagnetically-bound swarm of electrons. The chemical elements are distinguished from each other by the number of protons that are in their atoms.
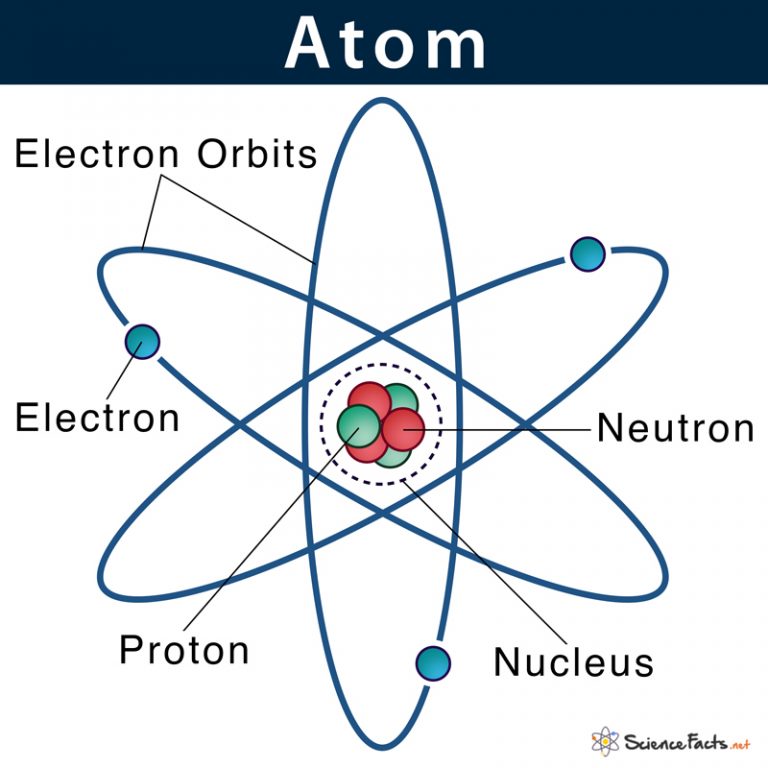
Atom Definition, Structure & Parts with Labeled Diagram
Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74). Since the iodine is added as a 1− anion, the number of electrons is 54 [53 - (1-) = 54]. Exercise 2.2.1 2.2. 1. An ion of platinum has a mass number of 195 and contains 74 electrons.

Atomic Structure Broad Learnings
In this video we cover the structure of atoms, what are subatomic particles, energy levels, and stable and reactive atoms.Transcript and notesAtomic structur.
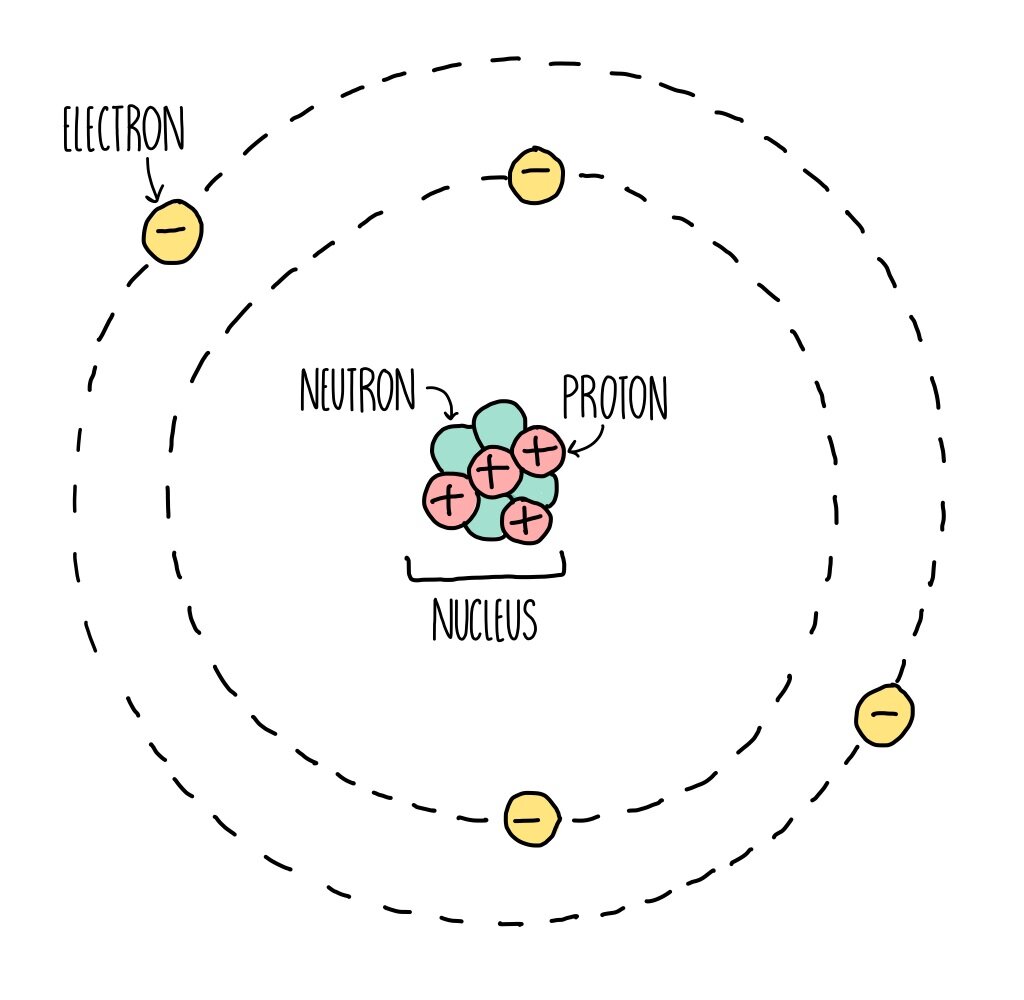
Atomic Structure (GCSE) — the science hive
Atom Diagram [/caption]The image on the left is a basic atom diagram. This one shows the protons, neutrons, and electrons of a carbon atom. Each is in a group of six. That makes the atom.
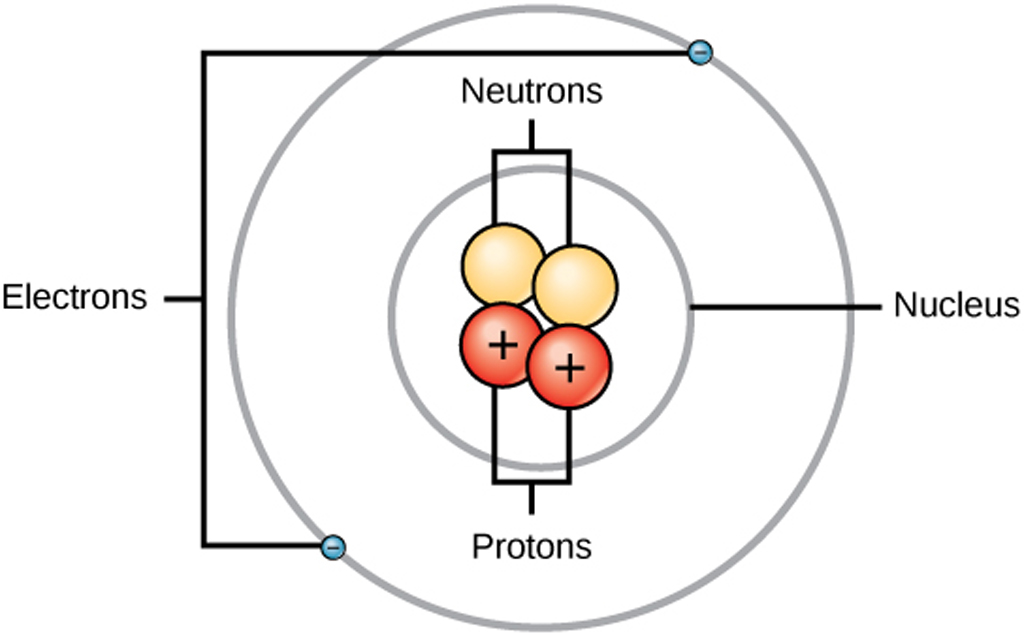
Structure of an Atom Structure & Use of Electron & Proton in Electronics
proton. Definition. Positively charged particle found in the nucleus of an atom. Location. Term. nucleus. Definition. The center of the atom which contains protons and neutrons. Location.
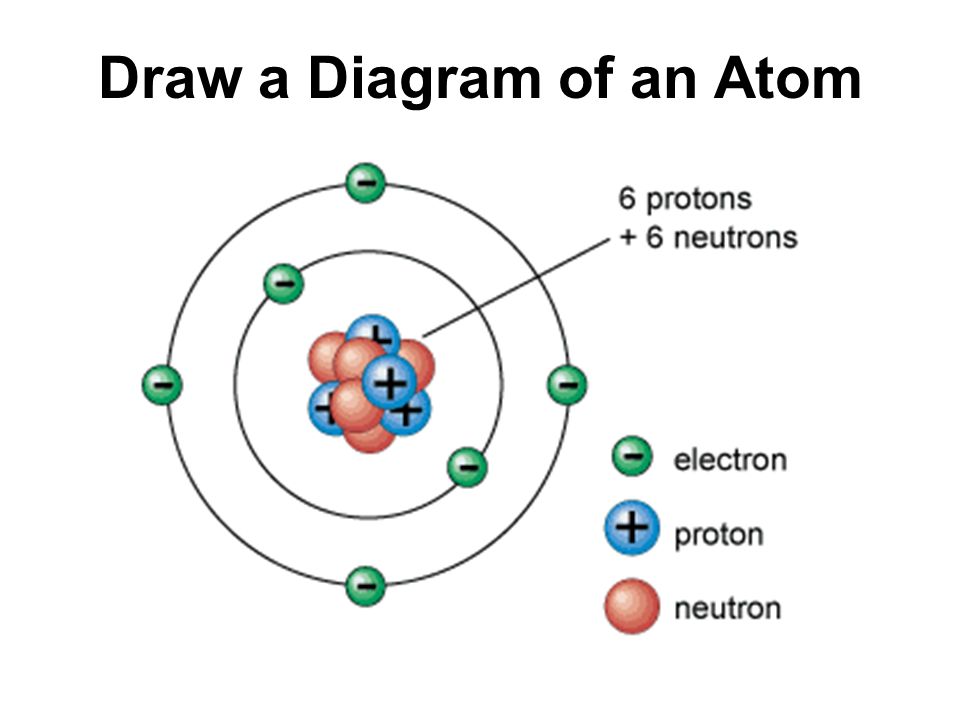
Skills Practice AMAZING 8TH GRADE SCIENTISTS
Our current model of the atom can be broken down into three constituents parts - protons, neutron, and electrons. Each of these parts has an associated charge, with protons carrying a positive.
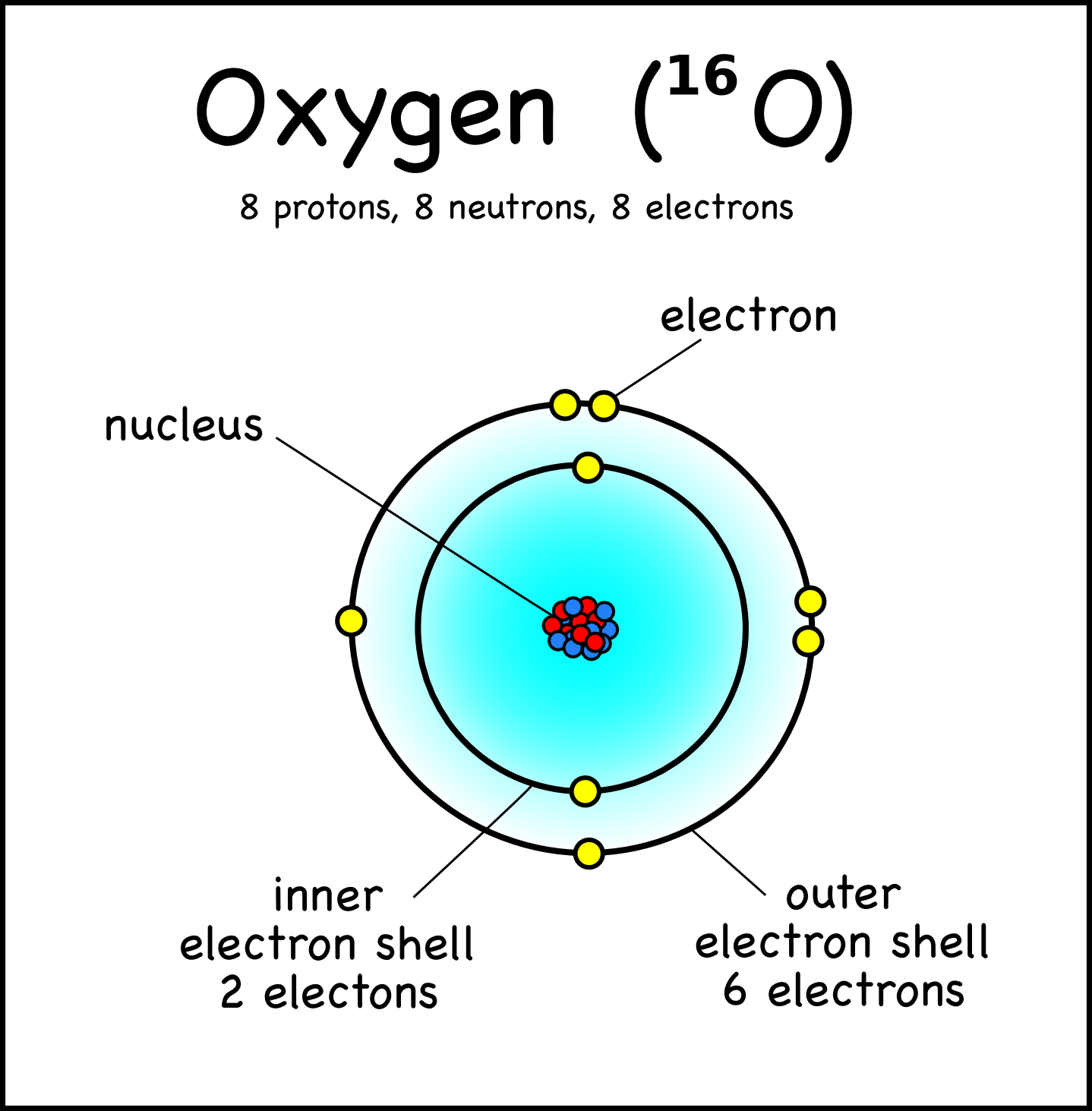
The Nucleus of the Atom and Radioactivity
Key Points. An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Protons and neutrons have approximately the same mass, about 1.67 × 10 −24 grams, which scientists define as one atomic.
/GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)
35 Label The Parts Of The Atom In The Diagram Below Labels For Your Ideas
1. Draw five protons in the nucleus of the atom. Label them with their charge. 2. Draw six neutrons in the nucleus of the atom. 3. Draw two electrons in the first energy level and label them with their charge. 4. Draw three electrons in the second energy level and label them with their charge. 5. What element is represented by the diagram?

Learn the Parts of an Atom
Atom: Definition, Structure & Parts with Labeled Diagram Atom Atoms are tiny particles that form the basic building blocks of all matter in the universe, whether solid, liquid, or gas. All living organisms and nonliving objects found on Earth are made of trillions and trillions of atoms.
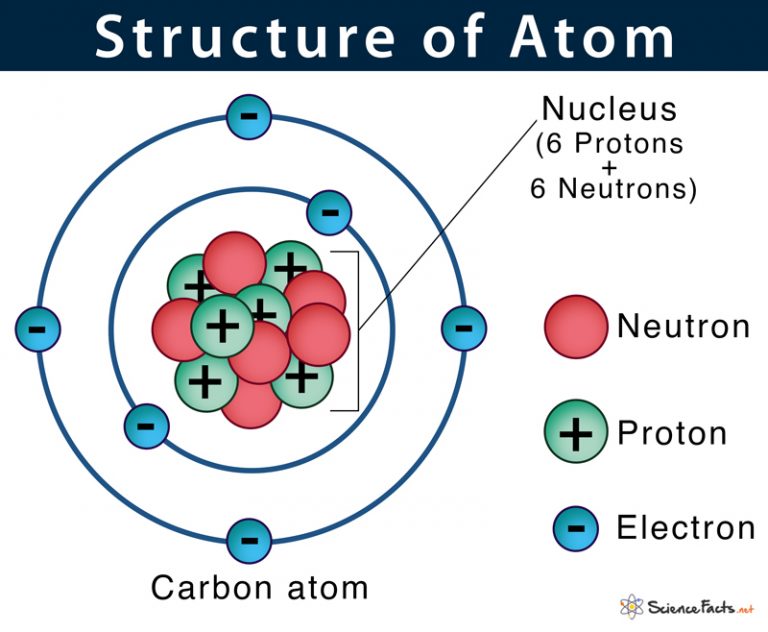
atom diagram to label
Mass number. It is total number of proton and neutron present in the nucleus of each atom of an element. Mass number = No. of proton + no of neutrons. = atomic number - no of neutron. For example: the mass number of fluorine is 19 and atomic number is 9. Thus the number of neutron in an atom of fluorine is 19-9 =10.
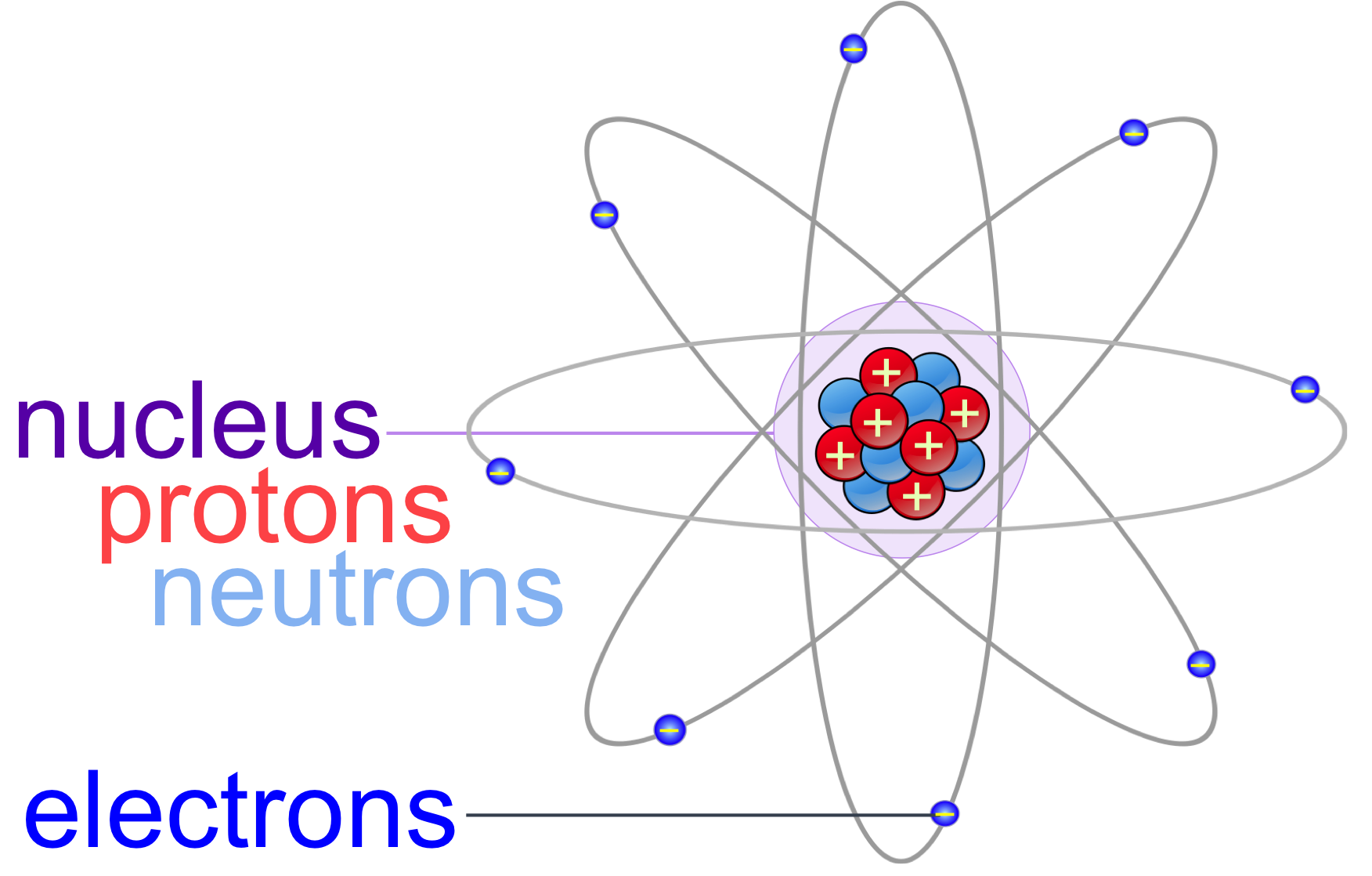
Atoms & Molecules echapter — The Biology Primer
The structure of a carbon atom, not drawn to scale The masses close mass The amount of matter an object contains. Mass is measured in kilograms (kg) or grams (g). of subatomic particles are very tiny.
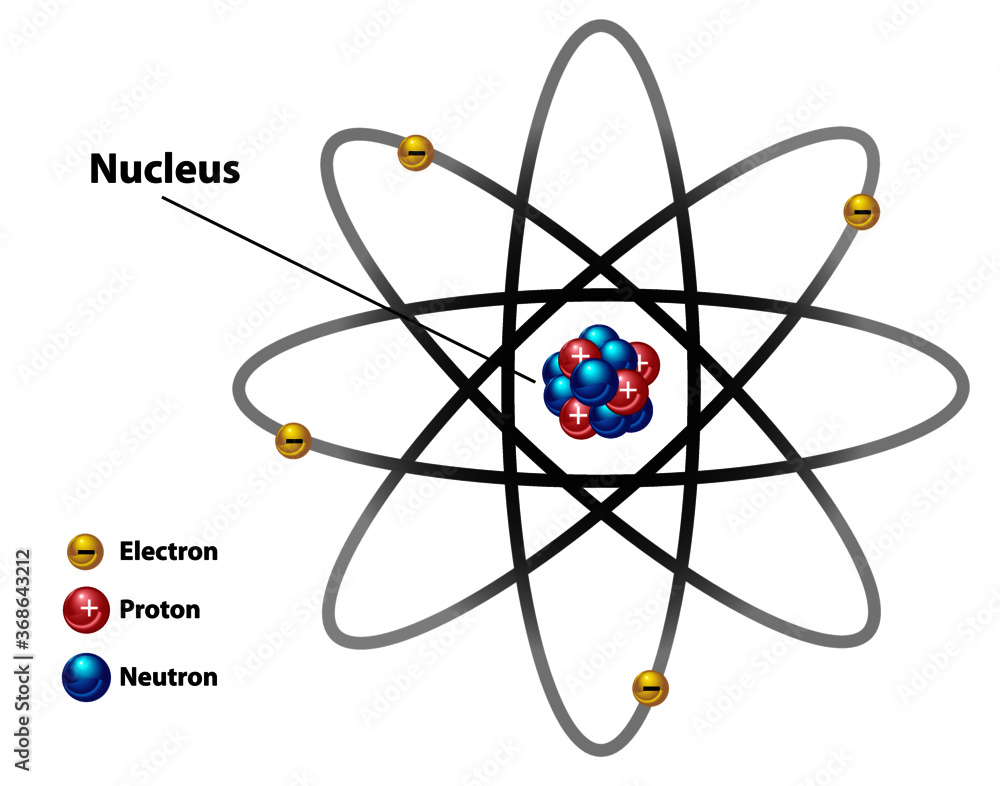
Atomic nucleus diagram labeled with electron, proton, and neutron. Stock Vector Adobe Stock
Parts of the Atom Part 1 - Label the parts of the atom below (protons, neutrons, electrons, nucleus, quarks). + + +----+ to the nucleus)? Part 2 - Answer the following questions. 1. _____What part of the atom has no charge? 2. _____What part of the atom has a positive charge? 3. _____What part of the atom has a negative charge? 4.